|
Do Now: On a whiteboard, draw the chemical equation for the combustion of methane with the correct balance of atoms on each side.
Task 1 Finish sharing the investigation results.
0 Comments
Lesson 6: How does bonding and breaking bonds affect energy in the system?
What have we figured out so far?
Do Now: Share with a table partner - one interesting thing that happened over your Spring Break. Warm up Kahoot - What are we learning about? (See Google Classroom to access the kahoot) Lesson:
3. Open the sim at this link (tinyurl.com/BondsandEnergyNetlogo) and play with the controls.
4. What does the simulation allow us to do that the marble model of bond making and breaking doesn’t? 5. What might be some limitations of this simulation as a model of what is going on when atoms rearrange in a chemical reaction? 6. Use the Discussion Diamond protocol to decide what questions we want to answer using the simulation.
8. Each group collaboratively PLANS their investigation. Exit ticket: What is your favorite Candy? Do Now: Image Connection
Find an image that represents something you have learned in chemistry. How creative can you get? Circle up with your images and whip around to share your connection with the class. Make sure you show the image to everyone as you explain your connection. Task 1: 10 minutes Check your review answers from yesterday using the key on Google Classroom (Mar 23). If you got an answer wrong, please confer with other students and have a conversation until you all understand the answer. I expect there to still be some confusion as we continue to figure this out. Task 2: You may use your notes and work, but please work alone.
Driving Question: Why does the breaking and making of bonds lead to temperature changes? How can we learn more about the energy in chemical reactions using the Netlego simulation?
Exit Ticket: "One Word". Please tell one word that sticks in your head from today. Do Now:
DRIVING QUESTION: Why does hydrogen fuel release more energy when burned that other fuels? Yesterday we used marbles and magnets to model the making and breaking of bonds. Today, please work with your group to step through the thinking of the parts of the model and how they represent the energy absorbed or released when bonds are broken and made. HANDOUT containing the thinking questions Watch this video to see the breaking bonds model. Exit ticket: Tell me one thing you now know you did not know before. Do Now: On a whiteboard, please list the missing words...
Attraction between two atoms will naturally lead to a(n) ____1____ in ___2___ energy within the reaction system. To break a bond between two atoms you must _____3_____ energy into/out of the reaction system to pull apart or knock off the atoms from each other. DRIVING QUESTIONS:
Navigate: Use a whiteboard
Do Now: Join our Quizlet Live using the code on the board. Quizlet Study Set link
Please use this Study Set to learn the vocabulary for future assessments and learning activities. DRIVING QUESTION: Could any chemical reaction be used to fuel a car? Do all chemical reactions ”release” energy? Please get a whiteboard and marker for each person.
Do Now: Please get out your notebooks.
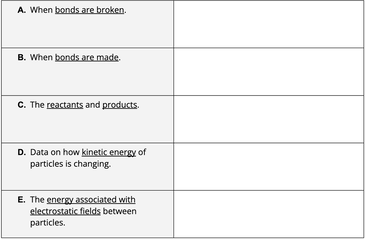
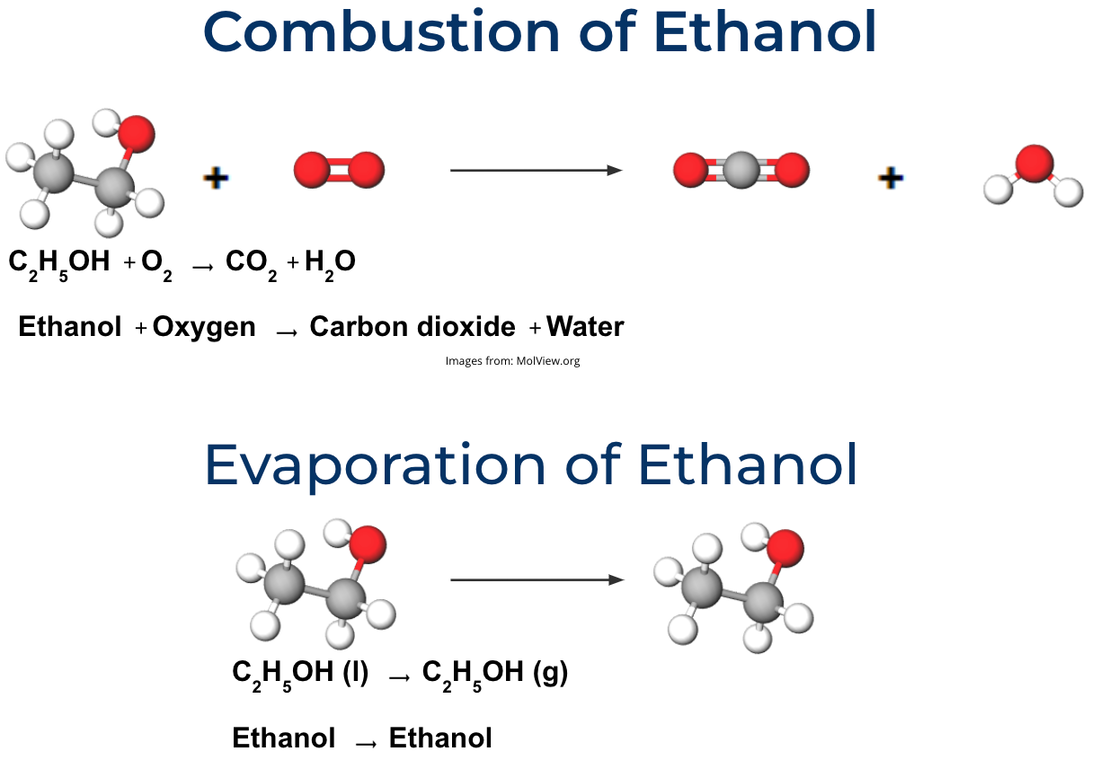
Concept focus: ENERGY and MATTER Let's model the energy transforming and transferring into and out of our system. LOL diagrams in your notebooks Last class, we were discussing questions we had and things we noticed about the combustion reactions. Let's pick up where we left off. What do you remember about our discussion? What is a fuel? What is necessary for something to be a combustion reaction? We create CO2 and water as well... Video forest fire. Do Now: At your table, make the reactants in the combustion reaction of ethanol. The bond with two rings represents a double bond. Burning ethanol - what happens to the matter and energy? How do we know?
Observations that could be evidence: What those observations mean: Make a model in your notebook at the molecular level of what is happening to the molecules during combustion of ethanol. We will do this together. How can we model the change in energy as ethanol combusts? LOL diagrams Learning Goals:
|
AuthorChris Campbell Science Practices, Core Ideas and Crosscutting ConceptsArchives
June 2023
|



 RSS Feed
RSS Feed
