|
A mole by any other name is not a gram.
Finish exploring and practicing with calculating the molecular weight of atoms and molecules.  Why does hydrogen give off more energy than octane per gram? How can we use a computational model to help figure this out? Lesson 8-9
 See Google Classroom for a practice Kahoot Finish Lesson 9 and Begin Lesson 10 What have we figured out so far? What new question do we have? What is the difference between grams and moles?
Begin Lesson 9 Driving Question: Can we explain why hydrogen has such a high energy output per gram compared to other fuels? Question 1-2: What are all of the pieces that we’ve figured out so far that would help us finally explain how it is that hydrogen produces so much more energy per gram than octane? Next Do now: Read this scientific statement and discuss to find what evidence we have for this:
Do You Know? What type of force is involved with atoms bonding? What questions did we propose from last lesson? NEXT: Grab the Lesson 8 Sheets and answer question #1 -
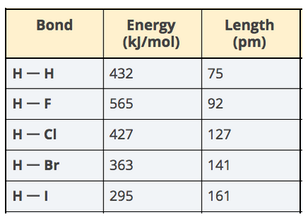
2. Data Analysis: Compile a Noticings and Wonderings chart. Use Supplement: Electron Shells and Bond Energy. 3. Model bond length: How can we improve the models we use to incorporate the 4. Watch video demonstrations of an inverter magnet and record quantitative and qualitative data to help figure out When is this system is most stable.
5. Thought Experiments 6. Building Understandings discussion Do Now: GET A WHITEBOARD AND MARKER
On a whiteboard, draw a graph that represents the energy changes during an endothermic reaction. You know this...energy at start, energy breaking bonds, energy making bonds. Our last driving question: "Why do some reactions give off heat and others take heat from the surroundings?" Make a model on your whiteboard showing an endothermic reaction and exothermic reaction. Include: Atoms, bonds, energy in, energy out, chemical reaction Task:
New Driving Question: What makes some bonds stronger and some bonds weaker? Why do atoms form bonds anyway? Task 2:
Monday:
Tuesday, Apr 18: Do Now:
Task 1: Review QUESTION 1: What happens to atoms and molecules in a chemical reaction (combustion, photosynthesis, etc...)
Most students gone on field trip.
Walk to the Lake and back and contemplate the chemistry of our world. Do Now: Answer this on a whiteboard:
If a chemical reaction gives off heat, are the bonds in the reactants or the products stronger? Unit Question: Why hydrogen fuel? Task 1: Fill out our model tracker for what we figured out in Lesson 6. Then we can skip the first three questions on their sheet. Task 2: See Google Classroom for your activity sheet for Lesson 7 Why doesn’t the energy that goes IN to break bonds and that comes OUT when bonds are formed just balance out? How might we investigate this question using the simulation? Make sure you are working as part of a group of at least three people. Choose who will work on which question and take 10 minutes to simulate the three different types of reactions. SMALL GROUP MODELING / EXPLANATIONS Use what you figured out in the last 4 investigations to explain and/or model on a separate sheet why the citric acid and sodium bicarbonate reaction resulted in a drop in temperature while burning fuel (either gasoline or ethanol) resulted in an increase in temperature. MODEL GALLERY WALK Post your model/explanation, then walk around the room to look at the other group’s work. Use sticky notes to leave feedback. After returning to your table, read through your group’s feedback and reflect on what you saw. What do you need to add or change about your model? |
AuthorChris Campbell Science Practices, Core Ideas and Crosscutting ConceptsArchives
June 2023
|




 RSS Feed
RSS Feed
