|
A mole by any other name is not a gram.
Finish exploring and practicing with calculating the molecular weight of atoms and molecules.
0 Comments
 Why does hydrogen give off more energy than octane per gram? How can we use a computational model to help figure this out? Lesson 8-9
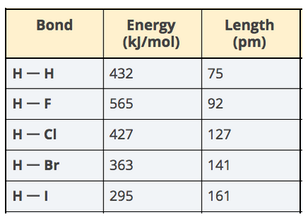 See Google Classroom for a practice Kahoot Finish Lesson 9 and Begin Lesson 10 What have we figured out so far? What new question do we have? What is the difference between grams and moles?
Begin Lesson 9 Driving Question: Can we explain why hydrogen has such a high energy output per gram compared to other fuels? Question 1-2: What are all of the pieces that we’ve figured out so far that would help us finally explain how it is that hydrogen produces so much more energy per gram than octane? Next |
AuthorChris Campbell Science Practices, Core Ideas and Crosscutting ConceptsArchives
June 2023
|



 RSS Feed
RSS Feed
